Научная группа профессора А.Ф. Хлебникова и профессора М.С. Новикова
STRAIN group: STrained Rings & Active Intermediates investigations
Научная группа профессора А.Ф. Хлебникова и профессора М.С. Новикова
Рабочие помещения: 4231
Телефоны: Александр Феодосиевич Хлебников, Михаил Сергеевич Новиков 8(812)428-93-44, лаборатория 8(812)428-93-84.
|
Хлебников Александр Феодосиевич доктор химических наук, профессор e-mail: a.khlebnikov@spbu.ru Читаемые лекционные курсы: “Стереохимия и конформационный анализ”, “Органический синтез” для магистров 1 курса, “Современные методы планирования органического синтеза” для студентов 4 курса. |
|
|
Новиков Михаил Сергеевич доктор химических наук, профессор e-mail: m.s.novikov@chem.spbu.ru Читаемые лекционные курсы: “Органическая химия” для студентов 3 курса |
|
Научные сотрудники:
|
Конев Александр Сергеевич кандидат химических наук, старший преподаватель e-mail: a.konev@spbu.ru Читаемые лекционные курсы: «Квантовая органическая химия» для студентов 4 курса. |
|
|
Ростовский Николай Витальевич кандидат химических наук, ассистент e-mail: n.rostovskiy@spbu.ru |
|
|
Томашенко Олеся Александровна кандидат химических наук, стажёр-исследователь e-mail: tomashenko.oles@gmail.com |
|
|
Галенко Алексей Викторович кандидат химических наук, стажёр-исследователь e-mail: a.galenko@chem.spbu.ru |
|
Аспиранты:
|
Лукьянов Даниил Александрович
e-mail: lda93@yandex.ru |
 |
|
Сметанин Илья Алексеевич
e-mail: ilyasmetanin@rambler.ru |
|
|
Галенко Екатерина Евгениевна e-mail: kalinka.kate@gmail.com |
|
|
Марьянов Алексей Николаевич e-mail: marjanov-alexei@yandex.ru |
|
Студенты:
|
Сахаров Павел Алексеевич
e-mail: paivel@inbox.ru |
|
|
Фунт Лия Дмитриевна
e-mail: lia.funt@gmail.com |
|
|
Пошехонов Игорь Сергеевич
e-mail: igor7057@mail.ru |
|
|
Рувинская Юлия Олеговна
e-mail: july_kz@mail.ru |
|
|
Стрельников Артём Александрович
e-mail: artstre-33@mail.ru |
|
|
Михайлов Кирилл Игоревич
e-mail: mikhaylovcyril@yandex.ru |
|
|
Агафонова Анастасия Викторовна
e-mail: agg_95@mail.ru |
|
|
Мосягин Иван Павлович e-mail: chipo44@mail.ru |
|
|
Смолко Наталья Сергеевна |
|
|
Чернышев Андрей |
|
|
Шакирова Фируза |
|
ОСНОВНЫЕ НАПРАВЛЕНИЯ ИССЛЕДОВАНИЙ:
1. Химия напряженных малых циклов
![]()
1a. Синтез и применение азиринов в качестве молекулярных строительных блоков для получения новых гетероциклических систем и лигандов.
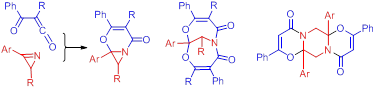
1b. Синтез и применение азиридинов в качестве эффективных источников активных интермедиатов для конструирования сложных молекулярных систем.
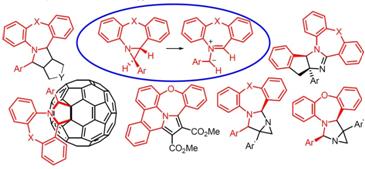
1с. Молекулярный дизайн хиральных триангуланов и синтез непредельных циклопропан-содержащих молекул с уникальными свойствами.

2. Новые активные интермедиаты как эффективный инструмент органического синтеза
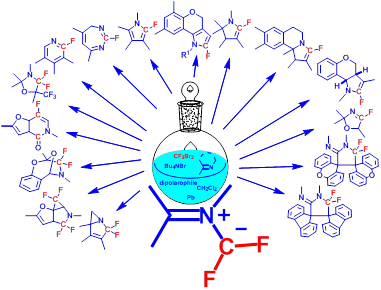
2a. Фторированные илиды азота: генерирование, свойства и применение в синтезе азот- и фтор-содержащих соединений.
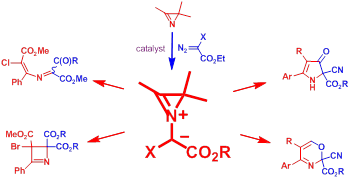
2b. Азириниевые илиды из азиринов, карбенов и диазосоединений: генерирование, свойства и применение в синтезе соединений с необычным сочетанием структурных фрагментов.
2c. Илидные и радикальные интермедиаты из азиридинов. Новые подходы и применение в гетероциклическом синтезе.
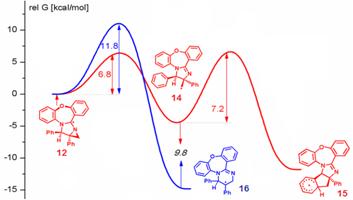
3. Направленный синтез молекул с полезными свойствами на основе напряженных молекул.
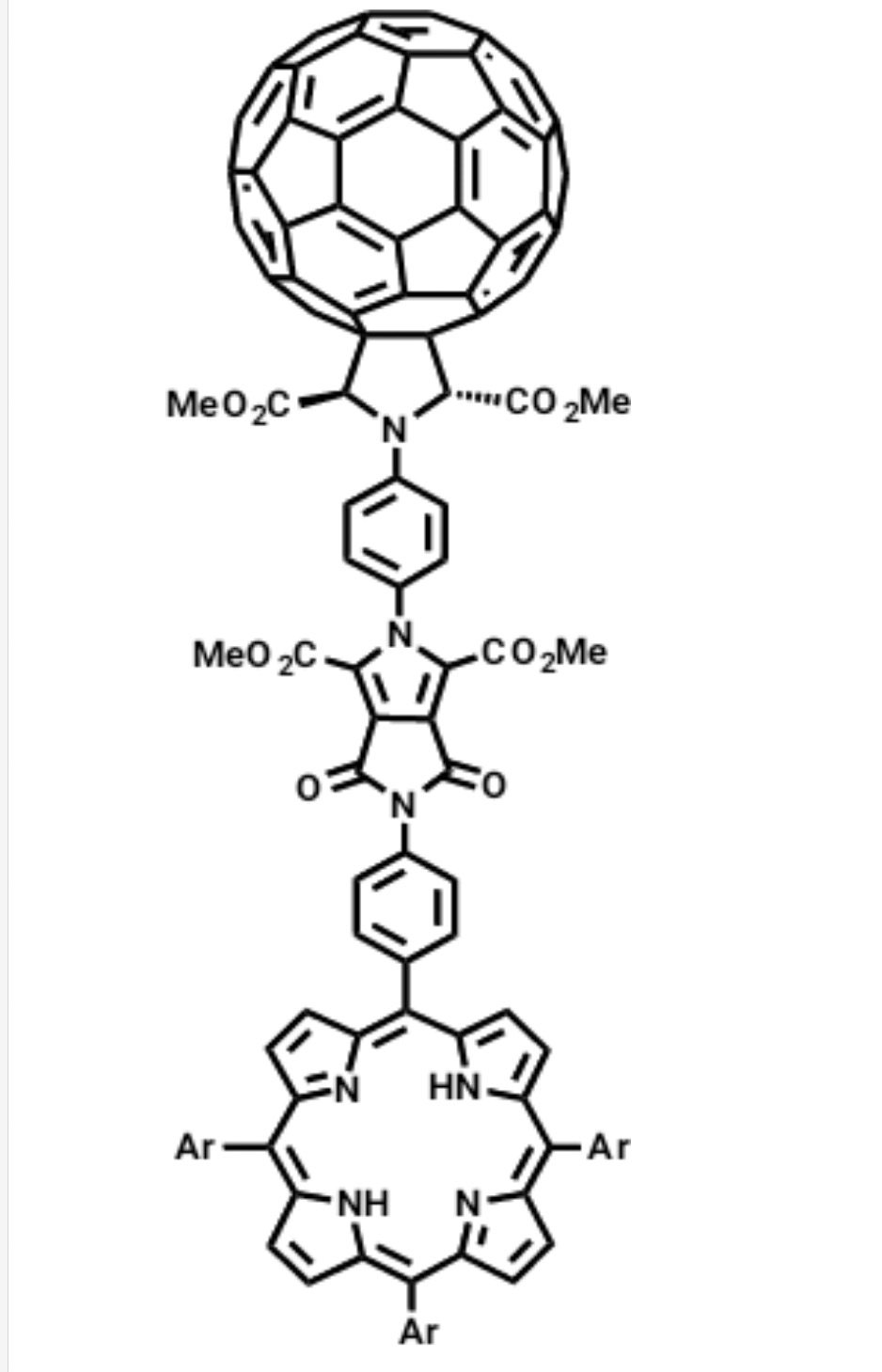
3a. Поиск новых подходов к синтезу наноразмерных фуллерен-содержащих систем для использования в фотовольтаике.
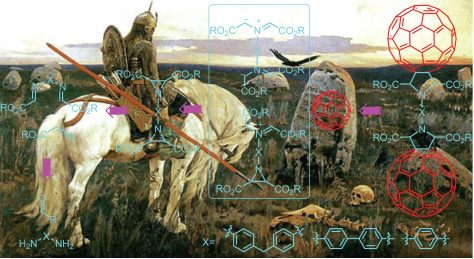
3b. Разработка методов синтеза новых фуллерен- и порфирин-содержащих молекул для фотовольтаики и искусственных фотосинтетических систем, а также порфирин-содержащих макромолекул с нелинейными оптическими свойствами.
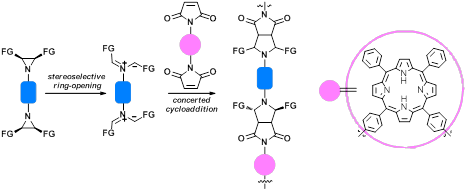
3с. Молекулярный дизайн новых азот-содержащих лигандов, включая NHC.
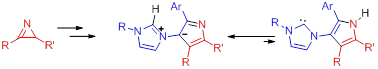
3d. Поиск путей синтеза и исследование новых фото- и термохромных соединений.
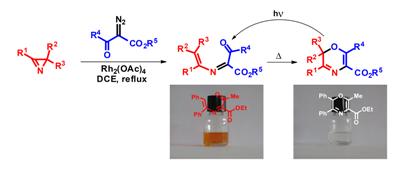
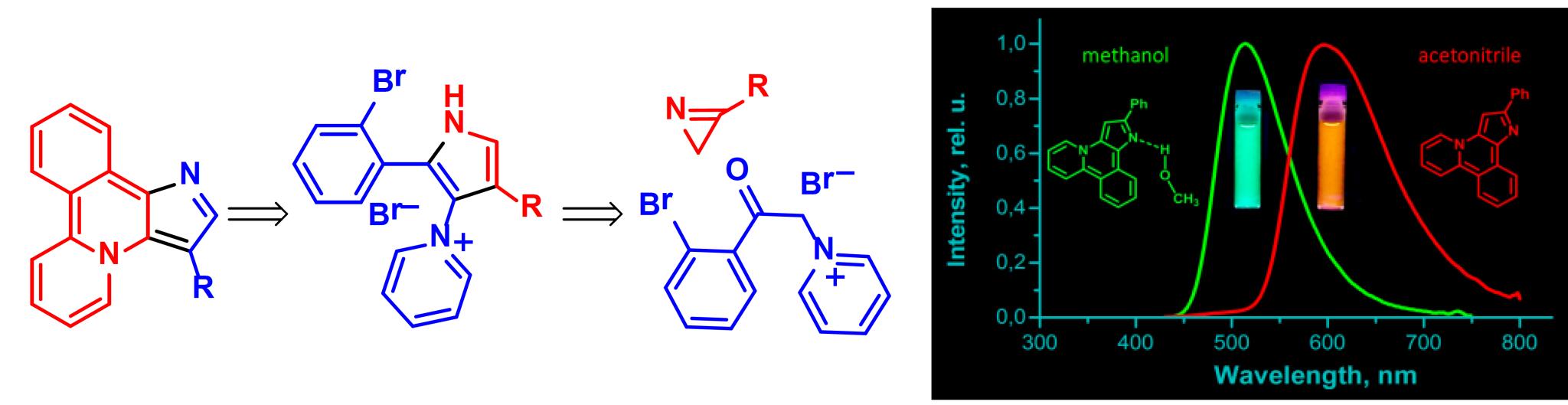
3e. Молекулярный дизайн новых полигетероциклических люминофоров.
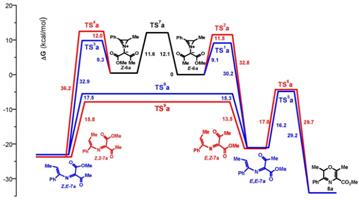
4. Квантово-химические расчеты для понимания механизмов реакций, реакционной способности интемедиатов и рационального поиска реакционных партнеров.
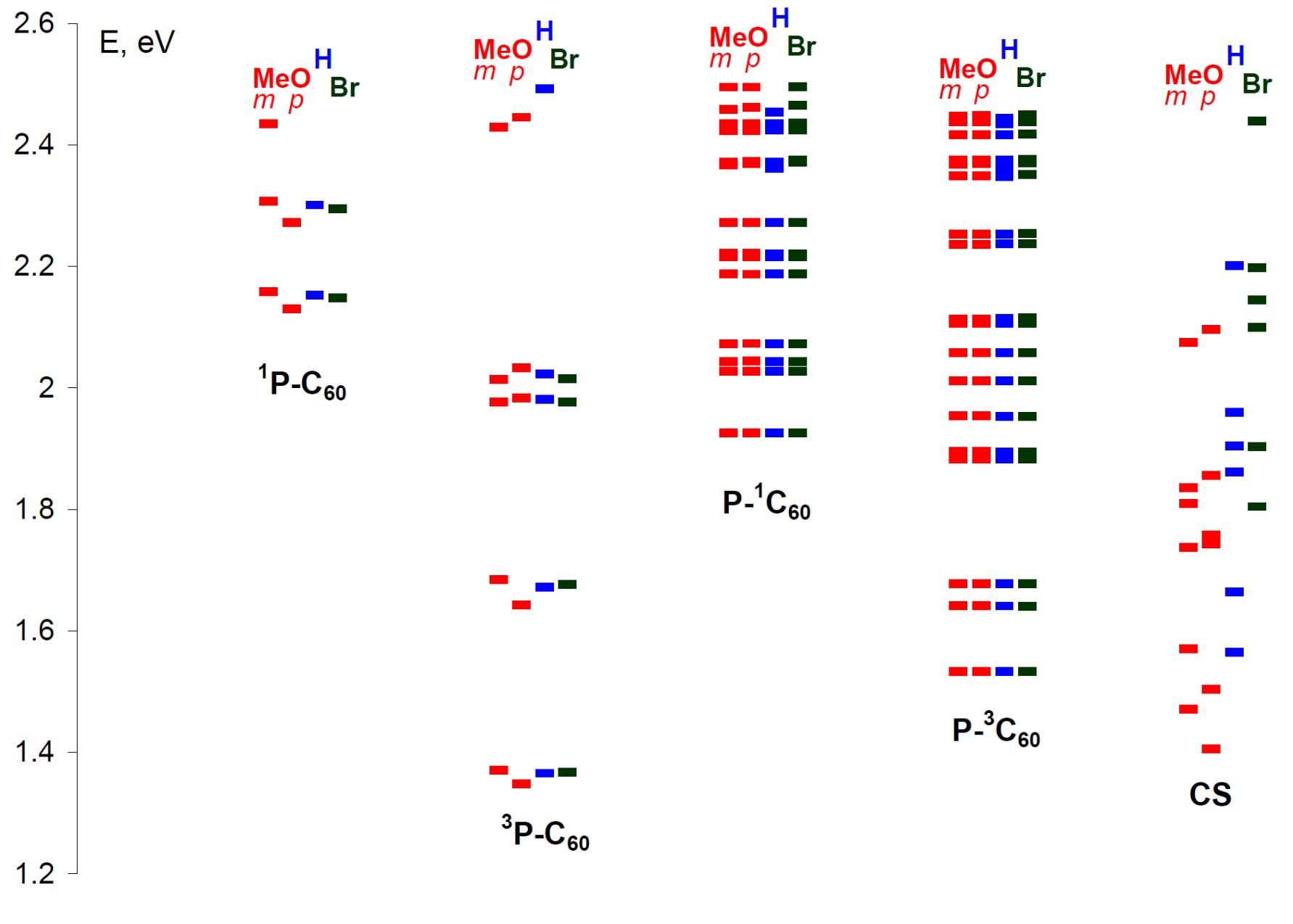
Ar = Ph, 4-BrC6H4, 4-MeOC6H4, 3-MeOC6H4
Избранные публикации:
List of publications.
Chapter:
| A. F. Khlebnikov, M. S. Novikov. Ring Expansions of Azirines and Azetines. Topics in Heterocyclic Chemistry, http://link.springer.com/chapter/10.1007/7081_2015_154. DOI: 10.1007/7081_2015_154, (Chapter in Synthesis of 4- To 7-Membered Heterocycles by Ring Expansion: Aza-, Oxa- And Thiaheterocyclic Small-Ring Systems, M.D. Hooghe,H.-J. Ha Eds, 2016, Springer; ISBN: 9783319249582; ISBN-10: 3319249584) |
 |
Papers:
-
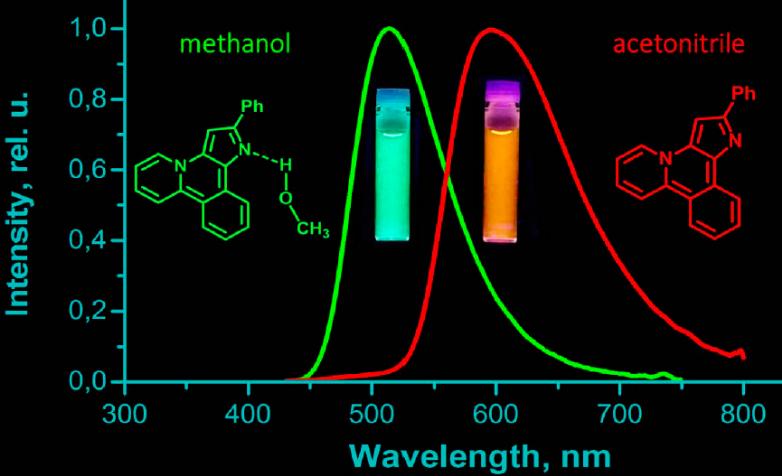
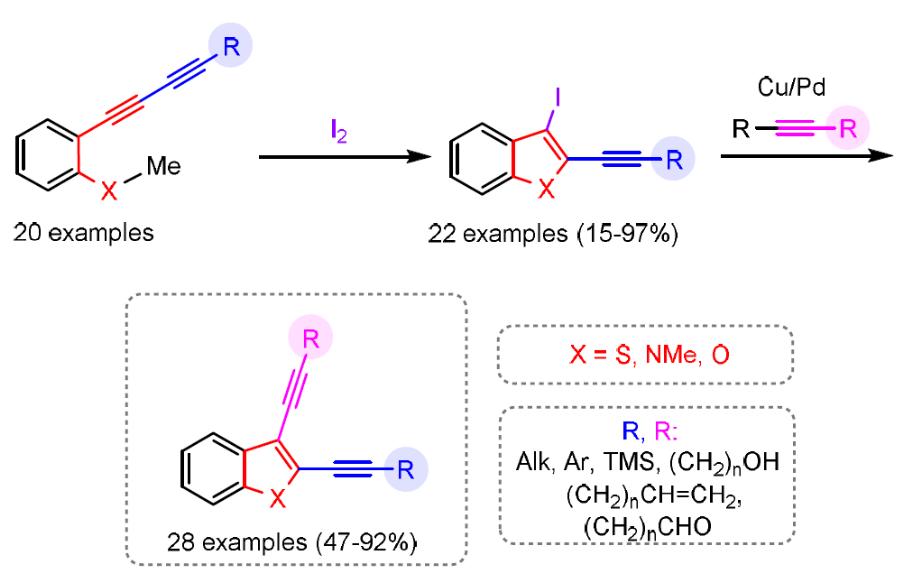
O. A. Tomashenko, A. F. Khlebnikov, I. P. Mosiagin, M. S. Novikov, E. V. Grachova, J. R. Shakirova, S. P. Tunik. “A new heterocyclic skeleton with highly tunable absorbtion/emission wavelength via H-bonding”. RSC Advances, 2015, 5, 94551-94561; DOI: 10.1039/C5RA17755C; http://pubs.rsc.org/en/content/articlehtml/2015/ra/c5ra17755c;
-
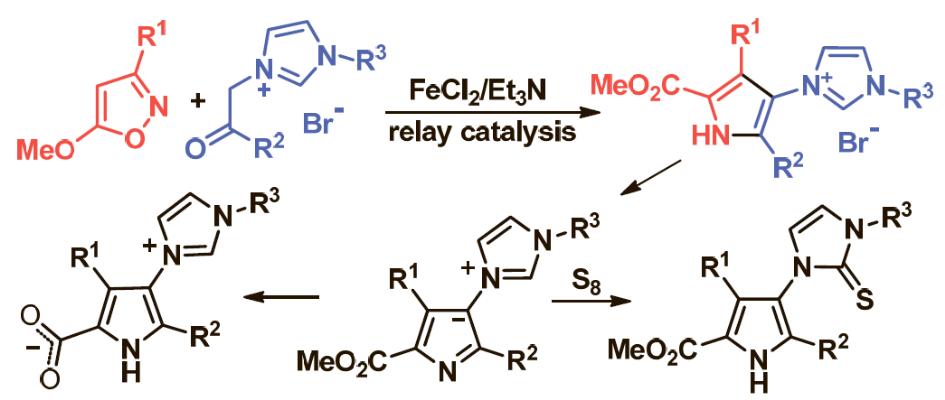
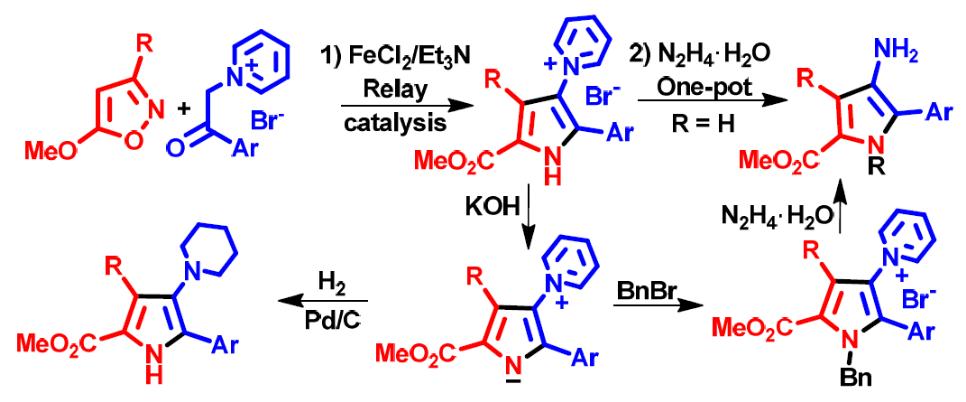
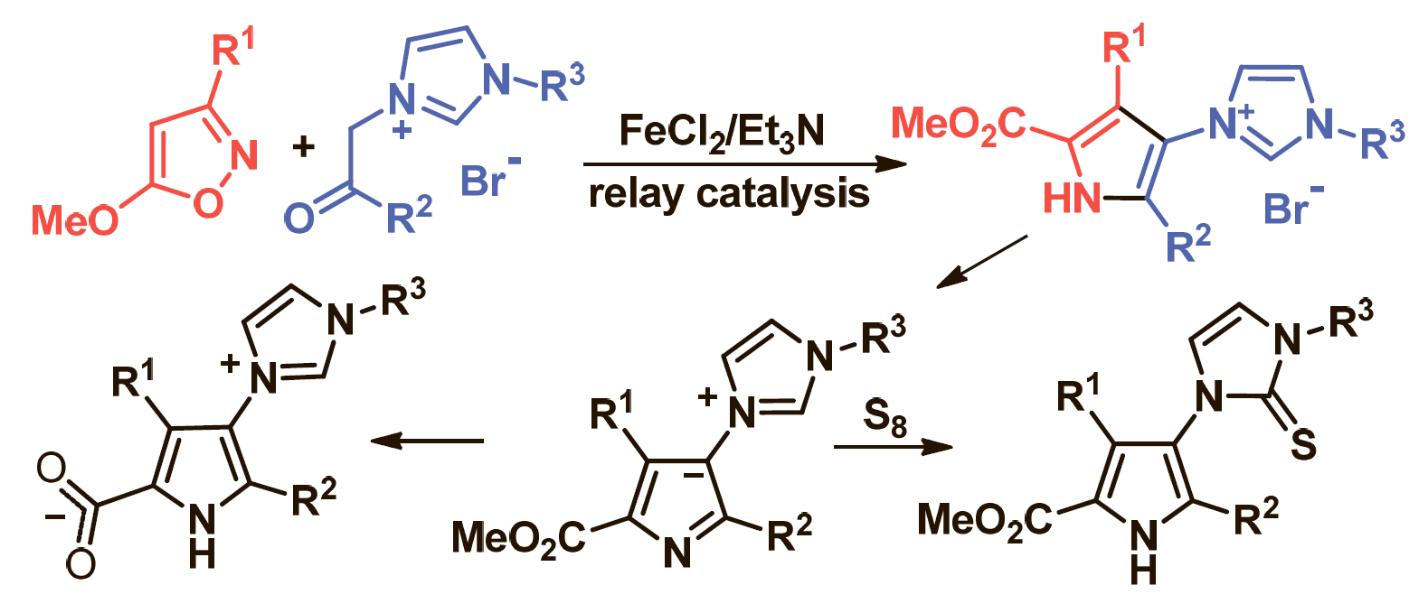
E. E. Galenko, O. A. Tomashenko, Khlebnikov A. F., Novikov M. S. “Fe(II)/Et3N-Relay-catalyzed domino reaction of isoxazoles with imidazolium salts in the synthesis of methyl 4-imidazolylpyrrole-2-carboxylates, its ylide and betaine derivatives”. Beilstein J. Org. Chem., 2015, 11, 1732-1740. DOI:10.3762/bjoc.11.189; http://www.beilstein-journals.org/bjoc/content/pdf/1860-5397-11-189.pdf;
- N. V.Rostovskii, P. A. Sakharov, M. S. Novikov, A. F. Khlebnikov, G. L. Starova. “Cu(I)-NHC-Catalyzed (2+3)-Annulation of Tetramic Acids with 2H-Azirines: Stereoselective Synthesis of Functionalized Hexahydropyrrolo[3,4-b]pyrroles”. Org. Lett., 2015, 17, 4148-4151; DOI: 10.1021/acs.orglett.5b01883; http://pubs.acs.org/doi/abs/10.1021/acs.orglett.5b01883;
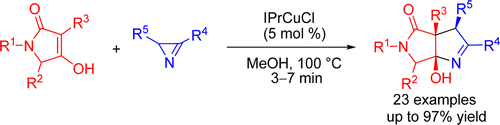
- E. E. Galenko, O. A. Tomashenko, A. F. Khlebnikov, M. S. Novikov. “Metal/organo relay catalysis in a one‐pot synthesis of methyl 4-aminopyrrole‐2‐carboxylates from 5‐methoxyisoxazoles and pyridinium ylides”. Org. Biomol. Chem., 2015, 13, 9825-9833; DOI: 10.1039/c5ob01537e; http://pubs.rsc.org/en/Content/ArticleLanding/2015/OB/c5ob01537e#!divAbstract;
- I. A. Smetanin, M. S. Novikov, N. V. Rostovskii, A. F. Khlebnikov, G. L. Starova, D. S. Yufit. “4-Halo-2-azabuta-1,3-dienes as intermediates in the rhodium carbenoid-initiated transformation of 2-halo-2H-azirines into 2,3-dihydroazetes and 2,5-dihydrooxazoles”. Tetrahedron, 2015, 71, 4616-4628. DOI: 10.1016/j.tet.2015.05.022, http://www.sciencedirect.com/science/article/pii/S0040402015006663;
-
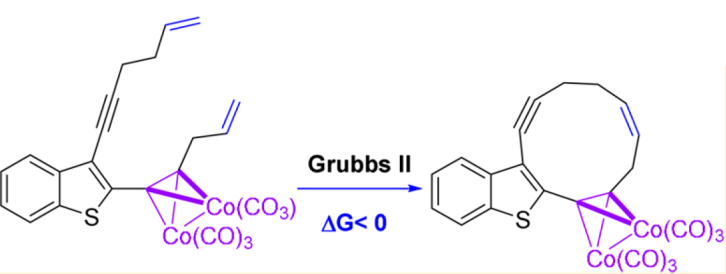
N. A. Danilkina, A. G. Lyapunova, A. F. Khlebnikov, G. L. Starova, S. Bräse, I. A. Balova “Ring-Closing Metathesis of Co2(CO)6–Alkyne Complexes for the Synthesis of 11-Membered Dienediynes: Overcoming Thermodynamic Barriers”. J. Org. Chem., 2015, 80, 5546-5555; DOI: 10.1021/acs.joc.5b00409; http://pubs.acs.org/doi/abs/10.1021/acs.joc.5b00409;
- Galenko A. V., Khlebnikov A. F., Novikov M. S., Pakalnis V. V., Rostovskii N. V. “Recent advances in isoxazole chemistry”. Russ. Chem. Rev., 2015, 84, 335–377; DOI: 10.1070/RCR4503; http://www.turpion.org/php/paper.phtml?journal_id=rc&paper_id=4503 http://www.turpion.org/php/paper.phtml?journal_id=rc&paper_id=4503;
- Galenko A. V., Khlebnikov A. F., Novikov M. S., Avdontseva M. S. “Synthesis of 3-(1,2-dioxoethyl)- and 2,3-dicarbonyl-containing pyrroles”. Tetrahedron, 2015, 71, 1940-1951; DOI: 10.1016/j.tet.2015.02.030; http://www.sciencedirect.com/science/article/pii/S0040402015001659;
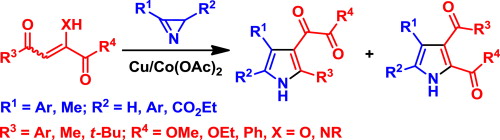
- Galenko E. E., Galenko A. V., Khlebnikov A. F., Novikov M. S. “Domino transformation of isoxazoles to 2,4-dicarbonylpyrroles under Fe/Ni relay catalysis”. RSC Advances, 2015, 5, 18172-18176; DOI: 10.1039/C5RA01889G; http://pubs.rsc.org/en/content/articlelanding/2015/ra/c5ra01889g#!divAbstract;
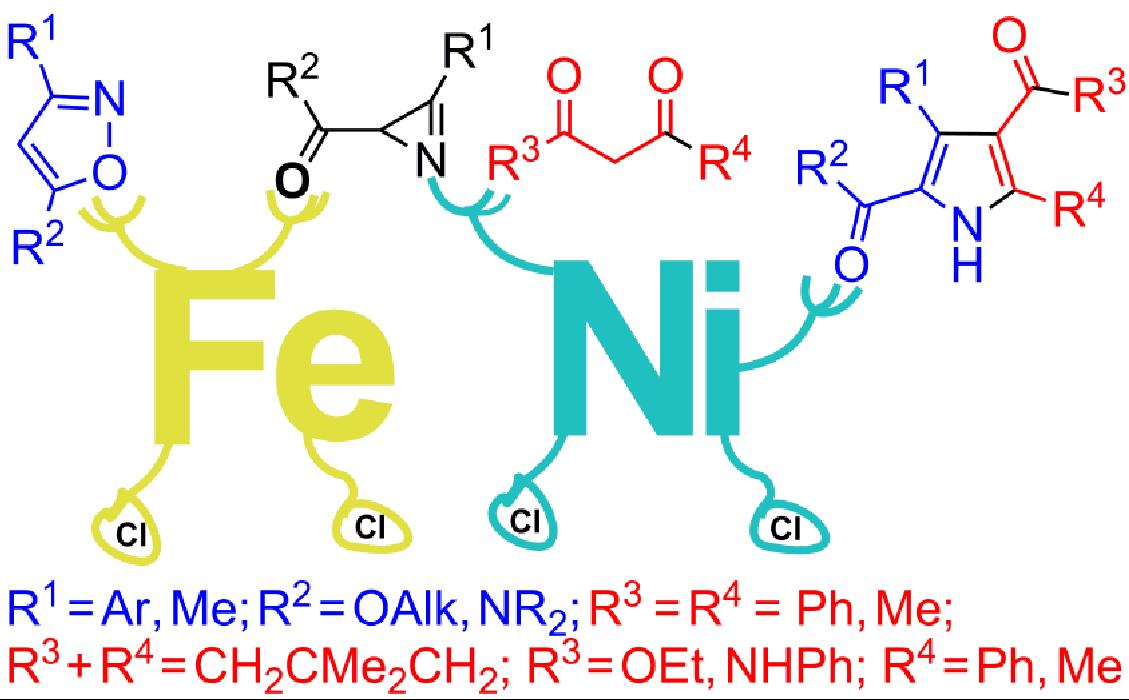
-
Rostovskii N. V., Novikov M. S., Khlebnikov A. F., Starova G. L., Avdontseva M. S. “Azirinium ylides from α-diazoketones and 2H-azirines on the route to 2H-1,4-oxazines: three-membered ring opening vs 1,5-cyclization”. Beilstein J. Org. Chem., 2015, 11, 302–312. DOI: 10.3762/bjoc.11.35, http://www.beilstein-journals.org/bjoc/single/articleFullText.htm?publicId=1860-5397-11-35;
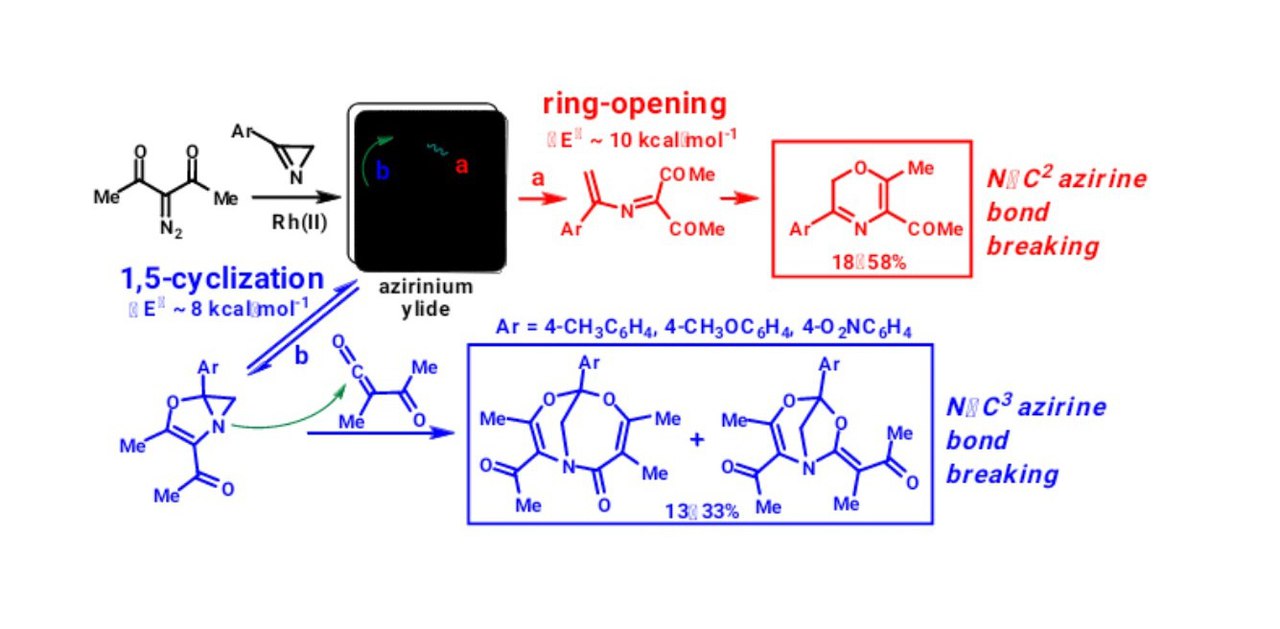
-
M. S. Novikov, A. F. Khlebnikov, N. V. Rostovskii, S. Tcyrulnikov, A. A. Suhanova, K. V. Zavyalov, D. S. Yufit. “Pseudopericyclic 1,5- versus pericyclic 1,4- and 1,6-electrocyclization in electron-poor 4-aryl-2-azabuta-1,3-dienes. Indole synthesis from 2H-azirines and diazo compounds”. J. Org. Chem., 2015, 80, 18-29. doi: 10.1021/jo501051n; http://pubs.acs.org/doi/abs/10.1021%2Fjo501051n;
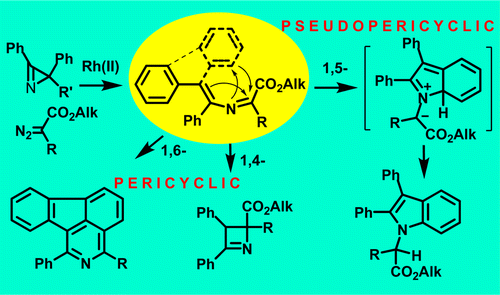
-
A. S. Konev, A. F. Khlebnikov, P. I. Prolubnikov, A. S. Mereshchenko, A. V. Povolotskiy, O. V. Levin, A. Hirsch. ”Synthesis of New Porphyrin–Fullerene Dyads Capable of Forming Charge-Separated States on a Microsecond Lifetime Scale”. Chem. Eur. J., 2015, 21, 1237-1250; DOI: 10.1002/ chem.201404435; http://onlinelibrary.wiley.com/doi/10.1002/chem.404435/abstract.
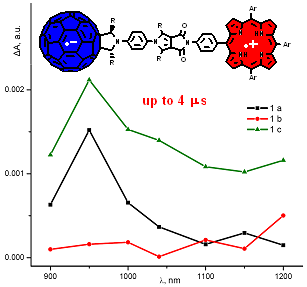
-
N. A. Danilkina, A. E. Kulyashova, A. F. Khlebnikov, S. Bräse, I. A. Balova. “Electrophilic Cyclization of Aryldiacetylenes in the Synthesis of Functionalized Enediynes Fused to a Heterocyclic Core”. J. Org. Chem., 2014, 79, 9018-9045; DOI: 10.1021/jo501396s; http://pubs.acs.org/doi/abs/10.1021/jo501396s
-
A. F. Khlebnikov, M. S. Novikov, Y. G. Gorbunova, E. E. Galenko, K. I. Mikhailov, V. V. Pakalnis, M. S. Avdontceva. “Isoxazolium N-ylides and 1-oxa-5-azahexa-1,3,5-trienes on the way from isoxazoles to 2H-1,3-oxazines”. Beilstein. J. Org. Chem., 2014, 10, 1896–1905. DOI:10.3762/bjoc.10.197; http://www.beilstein-journals.org/bjoc/content/pdf/1860-5397-10-197.pdf?a=y
-
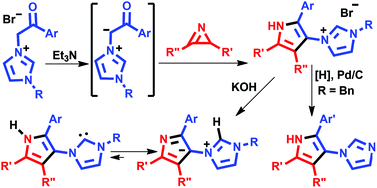
Khlebnikov, A. F.; Tomashenko, O. A.; Funt, L. D.; Novikov, M. S. “Simple Approach to Pyrrolylimidazole Derivatives by Azirine Ring Expansion with Imidazolium Ylides”. Org. Biomol. Chem., 2014, 12, 6598-6609; DOI: 10.1039/c4ob00865k; http://pubs.rsc.org/en/results?searchtext=DOI%3A10.1039%2Fc4ob00865k;
-
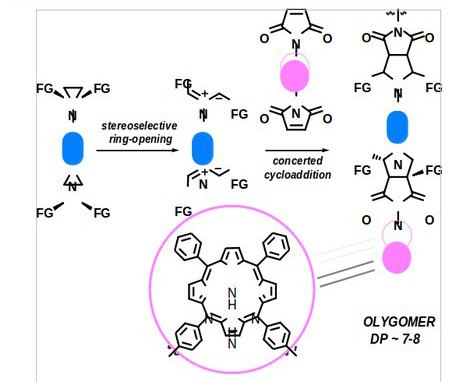
Konev, A. S., Lukyanov, D. A., Vlasov, P. S., Levin, O. V., Virtsev, A. A., Kislyakov, I. M.; Khlebnikov, A. F. “The Implication of 1,3-Dipolar Cycloaddition of Azomethine Ylides to the Synthesis of Main-Chain Porphyrin Oligomers”. Macromol. Chem. Phys., 2014, 215, 516-529. DOI: 10.1002/macp.201300679, http://onlinelibrary.wiley.com/doi/10.1002/macp.201300679/abstract;
-
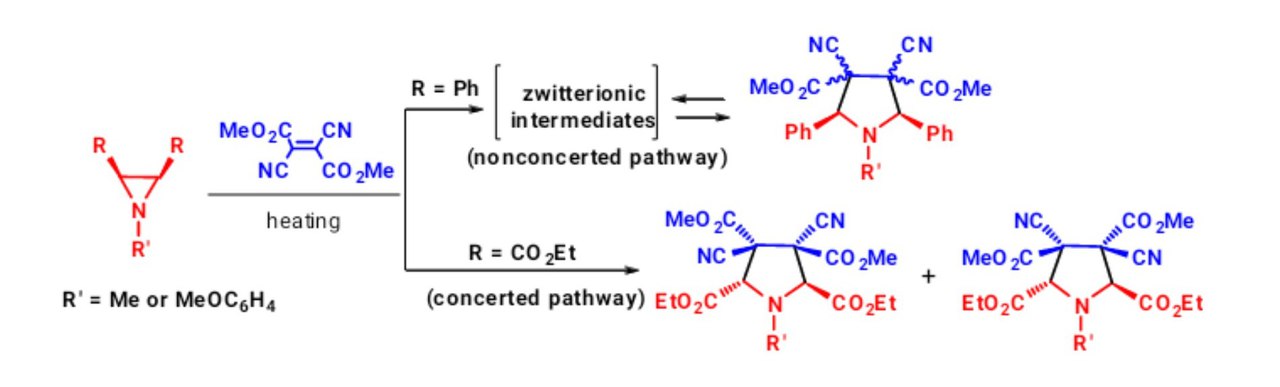
Khlebnikov, A. F., Konev, A. S., Virtsev, A. A., Yufit, D. S., Mlostoń, G., Heimgartner, H. “Concerted vs. Non-Concerted 1,3-Dipolar Cycloadditions of Azomethine Ylides to Electron-Deficient Dialkyl 2,3-Dicyanobut-2-enedioates”. Helv. Chim. Acta., 2014, 97, 453-470. DOI: 10.1002/hlca.201300405; http://onlinelibrary.wiley.com/doi/10.1002/hlca.201300405/abstract;
-
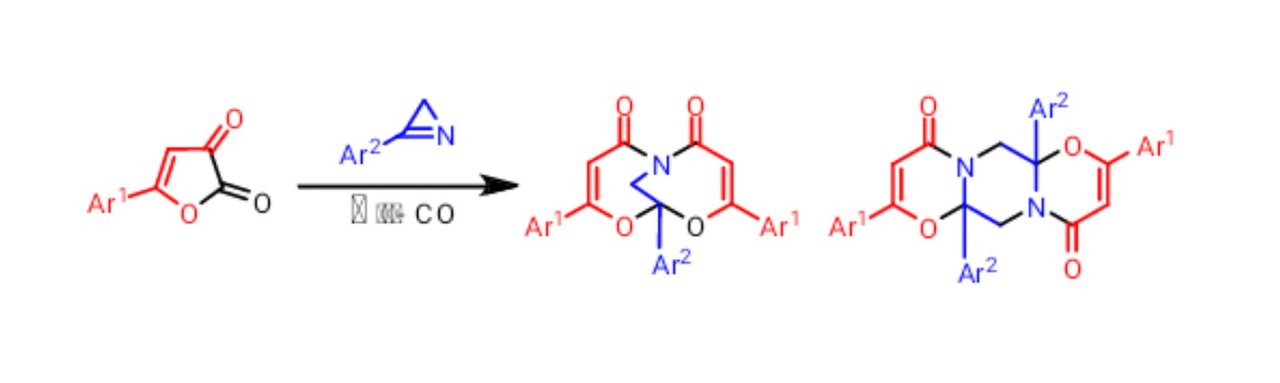
Khlebnikov, A. F.; Novikov, M. S.; Pakalnis, V. V.; Iakovenko, R. O.; Yufit D. S. “Domino reactions of 2H-azirines with acylketenes from furan-2,3-diones: Competition between the formation of ortho-fused and bridged heterocyclic systems”. Beilstein J. Org. Chem., 2014, 10, 784-793. DOI:10.3762/bjoc.10.74; http://www.beilstein-journals.org/bjoc/single/articleFullText.htm?vt=f&publicId=1860-5397-10-74&tpn=0&bpn=home;
-
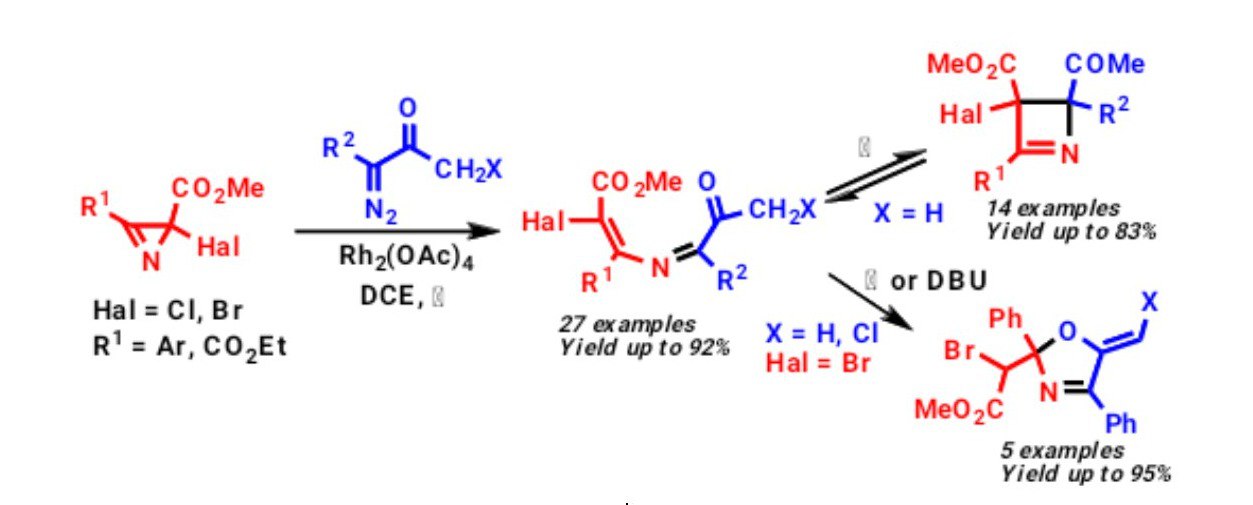
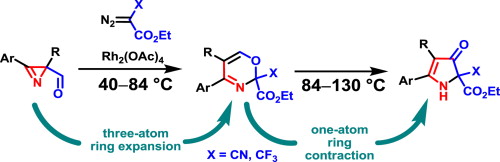
K. V. Zavyalov, M. S. Novikov, A. F. Khlebnikov, V. V. Pakalnis. “Selective syntheses of 2H-1,3-oxazines and 1H-pyrrol-3(2H)-ones via temperature-dependent Rh(II)-carbenoid-mediated 2H-azirine ring expansion”. Tetrahedron, 2014, 70, 3377-3384; DOI: 10.1016/j.tet.2014.03.101; http://www.sciencedirect.com/science/article/pii/S0040402014004633
-
A. S. Komolov, E. F. Lazneva, N. B. Gerasimova, A. A. Gavrikov, A. E. Khlopov, S. N. Akhremchik, M. V. Zimina, Yu. A. Panina, A. V. Povolotskii, A. S. Konev, A. F. Khlebnikov. “Electronic properties of ultrathin films based on pyrrolofullerene molecules on the surface of oxidized silicon”. Phys. Solid State, 2014, -56, 1659-1663; DOI: 10.1134/S1063783414080125; http://link.springer.com/article/10.1134/S1063783414080125;
-
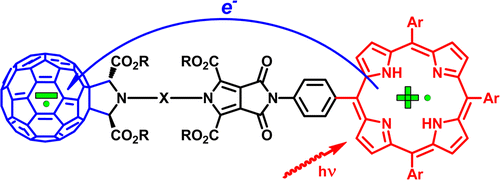
A. S. Konev, A. F. Khlebnikov, T. G. Nikiforova, A. A. Virtsev, H. Frauendorf. “Synthesis and spectroscopic and electrochemical properties of an axially symmetric fullerene-porphyrin dyad with a rigid pyrrolo[3,4-c]pyrrole spacer”. J. Org. Chem., 2013, 78, 2542-2552; DOI: 10.1021/jo302763a; http://pubs.acs.org/doi/abs/10.1021/jo302763a;
-
N. V. Rostovskii, M. S. Novikov, A. F. Khlebnikov, V. A. Khlebnikov, S. M. Korneev. “Rh(II)-carbenoid mediated 2H-azirine ring-expansion as a convenient route to non-fused photo- and thermochromic 2H-1,4-oxazines”. Tetrahedron, 2013, 69, 4292-4301; DOI: 10.1016/j.tet.2013.03.106: http://pubs.acs.org/doi/abs/10.1021/jo302763a;
-
K. V. Zavyalov, M. S. Novikov, A. F. Khlebnikov, D. S. Yufit. “Rh2(OAc)4-catalyzed reaction of α-diazocarbonyl compounds with 2-carbonyl-substituted 2H-azirines”. Tetrahedron, 2013, 69, 4546-4551; DOI: 10.1016/j.tet.2013.04.022; http://pubs.acs.org/doi/abs/10.1021/jo302763a;
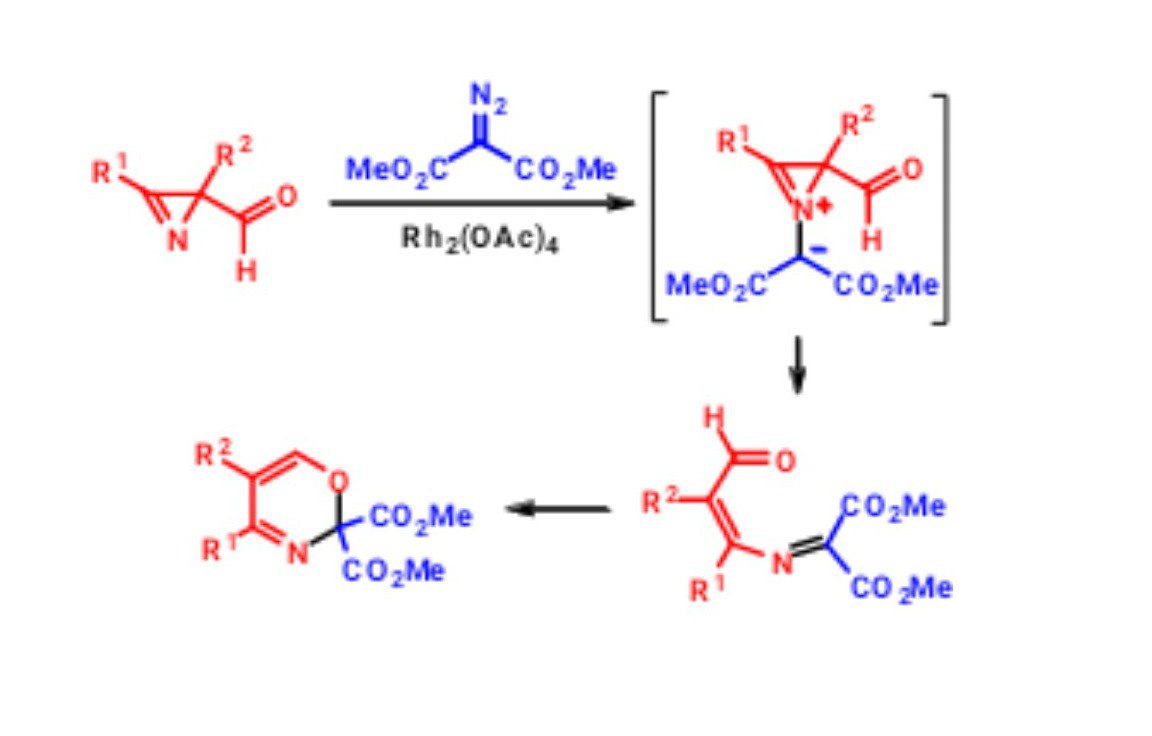
-
N. V. Rostovskii, M. S. Novikov, A. F. Khlebnikov, S. M. Korneev, D. S. Yufit. “Cu(II)-catalyzed domino reaction of 2H-azirines with diazotetramic and diazotetronic acids. Synthesis of 2-substituted 2H-1,2,3-triazoles”. Org. Biomol. Chem., 2013, 11, 5535-5545; DOI: 10.1039/c3ob40708j; http://pubs.rsc.org/en/Content/ArticleLanding/2013/OB/c3ob40708j#!divAbstract;
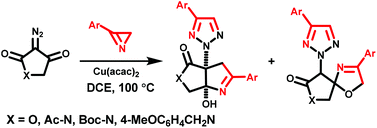
-
A. F. Khlebnikov, M. S. Novikov. „Recent advances in 2H-azirine chemistry”. Tetrahedron, 2013, 69, 3363-3401; DOI: 10.1016/j.tet.2013.02.020; http://www.sciencedirect.com/science/article/pii/S0040402013002123;
-
M. Arndt, G. Hilt, A. F. Khlebnikov, S. I. Kozhushkov, A. de Meijere. “Diels–Alder Reactions for the Construction of Cyclopropylarenes”. Eur. J. Org. Chem., 2013, 1171–1172; DOI: 10.1002/ejoc.201201724; http://onlinelibrary.wiley.com/doi/10.1002/ejoc.201201724/pdf;
-
A. S. Konev, A. A. Mitichkina, A. F. Khlebnikov, H. Frauendorf. “Stereoselective synthesis of cis- and trans-2′,5′-disubstituted N-arylpyrrolo[3′,4′:1,9](C60-I h )[5,6]fullerenes by the 1,3-dipolar cycloaddition of azomethine ylides from dialkyl aziridinedicarboxylates to fullerene C60”. Russ. Chem. Bull., 2012, 863-870; DOI: 10.1007/s11172-012-0121-7; http://link.springer.com/article/10.1007/s11172-012-0121-7; impact factor 0.423.
-
A. F. Khlebnikov, M. S. Novikov. „Fused aziridines as sources of azomethine ylides”. Chem. Heterocycl. Compd., 2012, 48, 179–190; DOI: 10.1002/chin.201233257; http://link.springer.com/article/10.1007%2Fs10593-012-0981-7; impact factor 0.634.
-
M. Arndt, G. Hilt, A. F. Khlebnikov, S. I. Kozhushkov, A. de Meijere. „Diels–Alder Reactions for the Construction of Cyclopropylarenes”. Eur. J. Org. Chem., 2012, 3112–3121; DOI: 10.1002/ejoc.201200105; http://onlinelibrary.wiley.com/doi/10.1002/ejoc.201200105/; impact factor 3.344.
-
A. F. Khlebnikov, S. I. Kozhushkov, D. S. Yufit, H. Schill, M. Reggelin, V. Spohr, A. de Meijere. „A Novel Type of Chiral Triangulane-Based Diphosphane Ligands for Transition Metals”. Eur. J. Org. Chem., 2012, 1530–1545; DOI: 10.1002/ejoc. 201101715; http://onlinelibrary.wiley.com/doi/10.1002/ejoc.201101715/abstract; impact factor 3.344.
-
A. F. Khlebnikov, M. V. Golovkina, M. S. Novikov, D. S. Yufit. “A novel strategy for the synthesis of 3-(N-heteryl)pyrrole derivatives”. Org. Lett., 2012, 14, 3768-3771; DOI: 10.1021/ol3016594; http://pubs.acs.org/doi/abs/10.1021/ol3016594; impact factor 6.142.
-
Kadina A. P., Khlebnikov A. F., Novikov M. S., J. Pérez P., Yufit D. S. “Intramolecular cycloaddition of azomethine ylides, from imines of O-acylsalicylic aldehyde and ethyl diazoacetate, to ester carbonyl – experimental and DFT computational study” Org. Biomol. Chem., 2012, 10, 5582-5591, DOI: 10.1039/c2ob25676b; http://pubs.rsc.org/en/Content/ArticleLanding/2012/OB/c2ob25676b#!divAbstract; impact factor 3.568.
-
Novikov M.S., Smetanin I.A., Khlebnikov A.F., Rostovskii N.V., Yufit D.S. “Synthesis of electron-poor 4-halo-2-azabuta-1,3-dienes by Rh(II)-catalyzed diazo ester-azirine coupling. 2-Azabuta-1,3-diene-2,3-dihydroazete valence isomerism”. Tetrahedron Lett., 2012, 53, 5777–5780; DOI: 10.1016/j.tetlet.2012.08.063; http://www.sciencedirect.com/science/article/pii/S0040403912014347; impact factor 2.397.
-
V. A. Rassadin, V. V. Sokolov, A. F. Khlebnikov, N. V. Ulin, S. I. Kozhushkov, A. de Meijere. “Convenient Synthesis of lexander Ethenylcyclopropane and Some 2-Cyclopropylcyclopropane Derivatives”. SYNTHESIS, 2012, 44, 372–376; DOI: 10.1055/s-0031-1289600; https://www.thieme-connect.com/products/ejournals/abstract/10.1055/s-0031-1289600; impact factor 2.5.
-
A. F. Khlebnikov, M. S. Novikov, M. V. Golovkina, P. P. Petrovskii, A. S. Konev, D. S. Yufit, H. Stoeckli-Evans. “An efficient approach to azirino and pyrrolo-fused dibenzazepines. Conformations of substituted dibenzo[c,f]pyrrolo[1,2-a]azepines”. Org. Biomol. Chem., 2011, 9, 3886-3895; DOI: 10.1039/C1OB05081H; http://pubs.rsc.org/en/content/articlelanding/2011/ob/c1ob05081h#!divAbstract; impact factor 3.568.
-
A. S. Konev, A. F. Khlebnikov, H. Frauendorf.“Bisaziridine tetracarboxylates as building blocks in the stereoselective synthesis of C60-fullerene diads and dumbbell-like bis-C60-fullerene triads”. J. Org. Chem., 2011, 76, 6218-6229; DOI: 10.1021/jo2009627; http://pubs.acs.org/doi/abs/10.1021/jo2009627; impact factor 4.564.
-
I. J. Kobylianskii, M. S. Novikov, A. F. Khlebnikov. „Formation and reactivity of gem-difluoro-substituted pyridinium ylides: Experimental and DFT investigation”. J. Fluor. Chem., 2011, 132, 175-180; DOI: 10.1016/j.jfluchem.2010.12.013; http://www.sciencedirect.com/science/article/pii/S0022113910003027; impact factor 1.939.
-
Khlebnikov A. F., Novikov M. S., Petrovskii P. P., Stoeckli-Evans H. “An Aza Cyclopropylcarbinyl-Homoallyl Radical Rearrangement–Radical Cyclization Cascade. Synthesis of Dibenzoimidazoazepine and Oxazepine Derivatives”. J. Org. Chem., 2011, 76, 5384–5391; DOI: 10.1021/jo200788k; http://pubs.acs.org/doi/abs/10.1021/jo200788k; impact factor 4.564.
-
S. I. Kozhushkov, A. F. Khlebnikov, R. R. Kostikov, D. S. Yufit, A. de Meijere. „Scalable synthesis of (1-cyclopropyl)cyclopropylamine hydrochloride”. Beilstein J. Org. Chem., 2011, 7, 1003–1006; DOI: 10.3762/bjoc.7.113; http://www.beilstein-journals.org/bjoc/single/articleFullText.htm?publicId=1860-5397-7-113; impact factor 2.801.
-
A. F. Khlebnikov, M. S. Novikov, V. V. Pakalnis, D. S. Yufit. “Non-concerted Cycloaddition of 2H-Azirines to Acylketenes – a Route to N-Bridgehead Heterocycles”. J. Org. Chem., 2011, 76, 9344–9352; DOI: 10.1021/jo201563b; http://pubs.acs.org/doi/abs/10.1021/jo201563b; impact factor 4.564.
-
A. F. Khlebnikov, M. S. Novikov, P. P. Petrovskii, A. S. Konev, D. S. Yufit, S. I. Selivanov, H. Frauendorf. Stereoselective Cycloaddition of Dibenzoxazepinium Ylides to Acetylenes and Fullerene C-60. Conformational Behavior of 3-Aryldibenzo[b,f]pyrrolo[1,2-d][1,4]oxazepine Systems. J. Org. Chem., 2010, 75, 5211-5215, DOI: 10.1021/jo100966j, http://pubs.acs.org/doi/abs/10.1021/jo100966j, импакт-фактор журнала: 4.638,
-
A. S. Konev, K. Abbaspour Tehrani, A. F. Khlebnikov, M. S. Novikov, J. Magull. Monofluorinated aziridines in asymmetric synthesis of chiral fluorinated prop-2-yn-1-amines. Russ. J. Org. Chem., 2010, 46, 976-986, DOI 10.1134/S1070428010070043, http://link.springer.com/article/10.1134/S1070428010070043, импакт-фактор журнала: 0.675
-
A. De Meijere, A. F. Khlebnikov, H. W. Sünnemann, D. Frank, K. Rauch, D. S. Yufit A. Convenient access to various 1-cyclopropylcyclopropane derivatives. Eur. J. Org. Chem., 2010, 3295-3301, DOI 10.1002/ejoc.201000209, http://onlinelibrary.wiley.com/doi/10.1002/ejoc.201000209/abstract, импакт-фактор журнала: 3.154,
-
A. F. Khlebnikov, M. S. Novikov. Strained Iminium Ylides. Russ. J. Gen. Chem., 2010, 80. 1652-1666, DOI 10.1134/S1070363210080153, http://link.springer.com/article/10.1134%2FS1070363210080153, импакт-фактор журнала: 0.64,
-
S. I. Kozhushkov, K. Wagner-Gillen, A. F. Khlebnikov, A. de Meijere. Productive Syntheses of 1-Ethynylcyclopropylamine and 1-Ethynylcyclobutylamine. Synthesis, 2010, 3967–3973, DOI 10.1055/s-0030-1258964, https://www.thieme-connect.de/DOI/DOI?10.1055/s-0030-1258964, импакт-фактор журнала: 2.443,
-
A. F. Khlebnikov, M. S. Novikov, P. P. Petrovskii, J. Magull, A. Ringe. Dibenzoxazepinium Ylides: Facile Access and 1,3-Dipolar Cycloaddition Reactions. Organic Lett., 2009, 11, 979-982, DOI 10.1021/ol802813a, http://pubs.acs.org/doi/abs/10.1021/ol802813a, импакт-фактор журнала: 6.324,
-
V. A. Khlebnikov, M. S. Novikov, A. F. Khlebnikov, N. V. Rostovskii. Rh(II)-Catalysed reactions of 2H-azirines with ethyl 2-acyl-2-diazoacetates. Synthesis of novel photochromic oxazines. Tetrahedron Lett., 2009, 50, 6509–6511, DOI 10.1016/j.tetlet.2009.09.033, http://www.sciencedirect.com/science/article/pii/S0040403909017432, импакт-фактор журнала: 2.391,
-
Kadina A.P., Novikov M.S., Khlebnikov A.F., Magull J. Azirino[c]imidazolyl Ylides in the Domino Reaction of 2,2-Dialkyl-4,6-diaryl-1,3-diazabicyclo[3.1.0]hex-3-enes with Dichlorocarbenes. Synthesis of (1RS,5SR,6RS)-4-(alk-1-enyl)-1,5-diaryl-3,7,7-trichloro-2,4-diazabicyclo[4.1.0]hept-2-enes. Chem. Heterocycl. Compd., 2008, 576-584, DOI: 10.1007/s10593-008-0077-6, http://link.springer.com/article/10.1007%2Fs10593-008-0077-6,
-
Novikov M. S., Khlebnikov A. F., Khistyaev K. A., Magull I. Isomerization and 1,3-dipolar cycloaddition of gem-difluorinated NH-azomethine ylides in the reaction of difluorocarbene with diarylmethanimines. Russ. Chem. Bull., 2008, 57, 1070-1079, DOI: 10.1007/s11172-008-0136-2, http://link.springer.com/article/10.1007%2Fs11172-008-0136-2,
-
Khistiaev K.A., Novikov M.S., Khlebnikov A.F., Magull J. gem-Difluorosubstituted nh-azomethine ylides in the synthesis of 4-fluorooxazolines via the three-component reaction of imines, trifluoroacetophenones and CF2Br2. Tetrahedron Lett., 2008, 49, 1237-1240, doi:10.1016/j.tetlet.2007.12.037, http://www.sciencedirect.com/science/article/pii/S0040403907024781,
-
Konev A. S., Stas S., Novikov M. S., Khlebnikov A. F., Abbaspour Tehrani K. Fluoroaziridines as novel substrates in the modified Petasis reaction: synthesis of monofluorinated propargyl amines. Tetrahedron, 2008, 64, 117-123, doi:10.1016/j.tet.2007.10.067, http://www.sciencedirect.com/science/article/pii/S0040402007018029,
-
Shinkevich E. Yu., Abbaspour Tehrani K., Khlebnikov A. F., Novikov M. S. Synthesis and reactivity of 3-(2-chloroalkyl)-2,2-dihaloaziridines. Tetrahedron, 2008, 64, 7524-7530, doi:10.1016/j.tet.2008.05.121, http://www.sciencedirect.com/science/article/pii/S0040402008010521,
-
Konev A. S., Khlebnikov A. F. A building block approach to monofluorinated organic compounds. Coll. Czech. Chem. Commun., 2008, 1553-1611, DOI: 10.1135/cccc20081553, http://dlib.lib.cas.cz/4002/,
-
Khlebnikov A.F., Novikov M.S., Dolgikh S.A., Magull J. 1,3- Vs. 1,5-Cyclization of azomethine ylides derived from 1-azabuta-1,3-dienes and difluoro- and dichlorocarbenes. Experimental and quantum-chemical study. Arkivoc, 2008, N9, 94-115, DOI: http://dx.doi.org/10.3998/ark.5550190.0009.909, http://quod.lib.umich.edu/a/ark/5550190.0009.909?view=pdf,
-
Konev A. S., Novikov M. S., Khlebnikov A. F., Abbaspour Tehrani K. A simple route to side-chain fluorinated beta-lactams from ring-fluorinated aziridines. J. Fluor. Chem., 2007, 128, P. 114-119, doi:10.1016/j.jfluchem.2006.10.013, http://www.sciencedirect.com/science/article/pii/S0022113906003976,
-
Shinkevich E.Yu., Novikov M.S., Khlebnikov A.F., Kostikov R.R., Kopf J., Magull J. New type of transannular reactions in azirine-fused medium-size heterocycles: selective transformations of azirino[2,1-e][1,6]benzoxazocines and -benzothiazocines into oxa(thia)zine and oxa(thia)zole derivatives. Russ. J. Org. Chem., 2007, 43, 1065-1079, DOI: 10.1134/S1070428007070214, http://link.springer.com/article/10.1134%2FS1070428007070214,
-
Konev, A.S., Novikov, M.S., Khlebnikov, A.F. Monofluoro-substituted azomethine ylides in fluorocarbene reactions with imines. Synthesis and transformations of monofluoroaziridines. Russ. J. Org. Chem., 2007, 43, 286-296, DOI: 10.1134/S1070428007020224, http://link.springer.com/article/10.1134%2FS1070428007020224,
-
Shinkevich E.Yu., Novikov M.S., Khlebnikov A.F. A convenient access to 3-(trihalomethyl)-3-phenyl-3,4-dihydro-2H-1,4- benzoxazines/thiazines and chlorinated 3-phenyl-2,3-dihydro-1,5-benzoxazepines/thiazepines by an aziridination-selective-ring-opening sequence. Synthesis, 2007, 225-230, DOI: 10.1055/s-2006-958938, https://www.thieme-connect.com/products/ejournals/abstract/10.1055/s-2006-958938,
-
Novikov M. S., Amer A. A., Khlebnikov A. F. Fluorinated 4H-1,3-diazepines by reaction of difluorocarbene with 2H-azirines. Tetrahedron Lett., 2006, 639-642, doi:10.1016/j.tetlet.2005.11.131, http://www.sciencedirect.com/science/article/pii/S0040403905026080,
-
Voznyi I. V., Novikov M. S., Khlebnikov A. F., Kostikov R. R. Reactions of 1,5-π-cyclization of gem-difluoro-substituted azomethine ylides involving an aromatic ring. Russ. J. Org. Chem., 2006, 42, 689-695, DOI: 10.1134/S1070428006050071, http://link.springer.com/article/10.1134%2FS1070428006050071,
-
Novikov M. S., Khlebnikov A. F., Egarmin M. A., Shevchenko M. V., Khlebnikov V. A., Kostikov R. R., Vidovic D. Regioselectivity of the 1,3-dipolar cycloaddition of fluorinated fluoren-9-iminium ylides to heteroelement-containing dipolarophiles: experimental and quantum-chemical study. Russ. J. Org. Chem., 2006, 42, 1800-1812, DOI: 10.1134/S1070428006120086, http://link.springer.com/article/10.1134%2FS1070428006120086,
-
Khlebnikov A. F., Novikov M. S., Amer A. A., Kostikov R. R., Magull J., Vidovic D. Azirinium ylides from alkoxycarbonylcarbenoids and 2H-azirines: Generation and transformations. Russ. J. Org. Chem., 2006, 42, 515-526, DOI: 10.1134/S1070428006040075, http://link.springer.com/article/10.1134%2FS1070428006040075,
-
Kusei E. Yu., Novikov M. S., Khlebnikov A. F. Reaction of fluoro(phenyl)carbene with schiff bases: synthesis of 2-fluoro-2-phenylaziridines. Russ. J. Gen. Chem., 2005, 75, 1643-1647, DOI: 10.1007/s11176-005-0480-y, http://link.springer.com/article/10.1007%2Fs11176-005-0480-y#/page-1,
-
Novikov M. S., Khlebnikov A. F., Voznyi I. V., Besedina O. V., Kostikov R. R. Intramolecular 1,3-dipolar cycloaddition of geminal difluoro azomethine ylides at multiple carbon-carbon bonds. Russ. J. Org. Chem., 2005, 41, 361-369, DOI: 10.1007/s11178-005-0171-5, http://link.springer.com/article/10.1007%2Fs11178-005-0171-5,
-
Khlebnikov A. F., Novikov M. S., Kostikov R. R., Kopf J. Intramolecular 1,3-dipolar cycloaddition of azomethine ylides generated from ethoxycarbonylcarbenoids and Schiff bases. Russ. J. Org. Chem., 2005, 41, 1341-1348, DOI: 10.1007/s11178-005-0344-2, http://link.springer.com/article/10.1007%2Fs11178-005-0344-2,
-
Khlebnikov A. F., Novikov M.S., Shinkevich E.Yu., Vidovic D. Selective transannular ring transformations in azirino-fused eight-membered O,N- or S,N-heterocycles. Org. Biomol. Chem., 2005, 3, 4040-4042, DOI: 10.1039/B512409C, http://pubs.rsc.org/en/content/articlelanding/2005/ob/b512409c#!divAbstract ,
-
Voznyi I. V., Novikov M. S., Khlebnikov A. F. Synthesis of 6,6-difluorocyclopropa[b]furo[2,3-c]pyrrole and 7-fluorofuro[3,2-c]pyridine derivatives via 1,5-electrocyclization of carbene-derived azomethine ylides. Synlett, 2005, 1006-1008, DOI: 10.1055/s-2005-864820, https://www.thieme-connect.com/products/ejournals/abstract/10.1055/s-2005-864820,
-
Khlebnikov A. F., Novikov M. S., Bespokoev A.A., Kostikov R. R., Kopf J., Starikova Z. A., Antipin M. Yu. New Tandem Reactions of Metal Carbenoids. Intermolecular Formation of Azomethine Ylide from Methyl 2-Diazo-2-phenylacetate and Schiff Base: Intramolecular 1,3-Dipolar Cycloaddition. Russ. J. Org. Chem., 2005, 41, 922-932, DOI: 10.1007/s11178-005-0267-y, http://link.springer.com/article/10.1007%2Fs11178-005-0267-y,
-
Konev A. S., Novikov M. S., Khlebnikov A. F. The first example of the generation of azomethine ylides from a fluorocarbene: 1,3-cyclization and 1,3-dipolar cycloaddition. Tetrahedron Lett., 2005, 46, 8337-8340, doi:10.1016/j.tetlet.2005.09.165, http://www.sciencedirect.com/science/article/pii/S0040403905021635,
-
Khlebnikov A. F., Voznyi I. V., Novikov M. S., Kostikov R. R. Intramolecular Cycloaddition of Geminal Dichloroazomethine Ylides to Multiple Carbon–Carbon Bonds. Russ. J. Org. Chem., 2005, 41, 560-566, DOI: 10.1007/s11178-005-0204-0, http://link.springer.com/article/10.1007%2Fs11178-005-0204-0,
-
Khlebnikov A. F., Novikov M. S., Kostikov R. R. Iminium Ylides from Carbenes and Carbenoids: Generation and Synthetic Applications. Russ. Chem. Rev., 2005; 74, 171-192, DOI: 10.1070/RC2005v074n02ABEH000973, http://www.turpion.org/php/paper.phtml?journal_id=rc&paper_id=973,
-
Novikov M. S., Khlebnikov A. F., Shevchenko M. V., Kostikov R. R., Vidovic D. 1,3-Dipolar cycloaddition of difluoro-substituted azomethine ylides. Synthesis and transformations of 2-fluoro-4,5-dihydropyrroles. Russ. J. Org. Chem., 2005, 41, 1496-1506, DOI: 10.1007/s11178-005-0373-x, http://link.springer.com/article/10.1007%2Fs11178-005-0373-x,
-
Khlebnikov, A. F.; Novikov, M. S.; Amer, A. A. Reaction of difluorocarbene with 2H-azirines: generation and transformations of strained azomethine ylides – aziriniodifluoromethanides. Russ. Chem. Bull., 2004, 1092-1101, DOI: 10.1023/B:RUCB.0000041305.50408.ed, http://link.springer.com/article/10.1023%2FB%3ARUCB.0000041305.50408.ed,
-
Khlebnikov A. F., Novikov M. S., Amer A. A. Reactions of 2H-azirines with carbenoids from diazo esters: transformations of novel azirinium ylides. Tetrahedron Lett., 2004, 46, 6003-6006, doi:10.1016/j.tetlet.2004.06.038, http://www.sciencedirect.com/science/article/pii/S0040403904012870,
-
Voznyi I. V.; Novikov M. S.; Khlebnikov A. F.; Kostikov R. R. Azomethine ylides derived from dichlorocarbene and O-acylsalicylaldehyde anils in the synthesis of 2,5-epoxy-2,3,4,5-tetrahydro-1,4-benzoxazepin-2-ones and 2-aminoethanols. Russ. Chem. Bull., 2004, 1087-1091, DOI: 10.1023/B:RUCB.0000041304.68814.83, http://link.springer.com/article/10.1023%2FB%3ARUCB.0000041304.68814.83,
-
Voznyi I. V.; Novikov M. S.; Khlebnikov A. F.; Kopf J.; Kostikov R. R. Intramolecular 1,3-Dipolar Cycloaddition to Ester Carbonyl of Azomethinylides Prepared from Aldimines and Difluorocarbene. Russ. J. Org. Chem., 2004, 40, 199-205, DOI: 10.1023/B:RUJO.0000034942.42778.e0, http://link.springer.com/article/10.1023%2FB%3ARUJO.0000034942.42778.e0,
-
Novikov M. S., Khlebnikov A. F., Egarmin M. A., Kopf J., Kostikov R. R. 1,3-dipolar cycloaddition of fluorinated azomethine ylides at the C=N bond. Russ. J. Org. Chem., 2004, 40, 1493-1499, DOI: 10.1007/s11178-005-0048-7, http://link.springer.com/article/10.1007%2Fs11178-005-0048-7,
-
Novikov M. S.; Khlebnikov A. F.; Shevchenko M. V. A facile carbene route to 2-fluoro-2-pyrrolines via fluorinated azomethine ylides. J. Fluor. Chem., 2003, 123, 177-181, doi:10.1016/S0022-1139(03)00116-7, http://www.sciencedirect.com/science/article/pii/S0022113903001167,
-
Khlebnikov A. F., Novikov M. S., Kusei E. Yu., Kopf J., Kostikov R. R. Cascade transformations of (2,2-diaryl-3,3-dichloroaziridin-1-yl)acetates. Russ. J. Org. Chem., 2003, 39, 559-573, DOI: 10.1023/A:1026068020111, http://link.springer.com/article/10.1023%2FA%3A1026068020111,
-
Novikov M. S.; Khlebnikov A. F.; Kostikov R. R. 1,3-Dipolar Cycloaddition of Azomethine Ylides Generated from Ketimines an Difluorocarbene to Symmetrically Substituted Olefins. Russ. J. Org. Chem., 2002, 38, 1647 – 1654, DOI: 10.1023/A:1022518319349, http://link.springer.com/article/10.1023%2FA%3A1022518319349,
-
Khlebnikov A. F.; Novikov M. S.; Amer A. A. Generation and cycloadditions of azirinium difluoromethanides - strained azomethine ylides. Tetrahedron Lett., 2002, 43, 8523-8525, doi:10.1016/S0040-4039(02)02076-2, http://www.sciencedirect.com/science/article/pii/S0040403902020762,
-
Novikov M. S., Voznyi I. V., Khlebnikov A. F., Kopf J., Kostikov R. R. Unprecedented 1,3-dipolar cycloaddition of azomethine ylides to ester carbonyl. J. Chem. Soc., Perkin Trans. 1, 2002, 1628-1630, DOI: 10.1039/B204464A, http://pubs.rsc.org/en/content/articlelanding/p1/2002/b204464a#!divAbstract,
-
Novikov M. S.; Khlebnikov A. F.; Sidorina E. S.; Masalev A. E.; Kopf J.; Kostikov,R. R. 1,3-Dipolar Cycloaddition of Azomethine Ylides Generated from Schiff Bases Difluorocarbene to Symmetric Olefins. Russ. J. Org. Chem., 2002, 38, 672 – 682, DOI: 10.1023/A:1019659021891, http://link.springer.com/article/10.1023%2FA%3A1019659021891,
-
Novikov M. S., Khlebnikov A. F., Besedina O. V., Kostikov R.R. First example of intramolecular cycloaddition of carbene-derived azomethine ylides in a domino reaction of difluorocarbene with Schiff bases. Tetrahedron Lett., 2001, 42, 533-535, doi:10.1016/S0040-4039(00)02105-5, http://www.sciencedirect.com/science/article/pii/S0040403900021055,
-
Khlebnikov A. F., Novikov M. S., Khlebnikov V. A., Kostikov R. R. Prototropic generation of azomethine ylides from esters of n-benzylideneaminoacids, and 1,3-dipolar cycloaddition thereof. Russ. J. Org. Chem., 2001, 37, 507-512, DOI: 10.1023/A:1012425701412, http://link.springer.com/article/10.1023%2FA%3A1012425701412,
-
Novikov M. S., Khlebnikov A. F., Sidorina E. S., Kostikov R. R. 1,3-Dipolar cycloaddition of azomethine ylides derived from imines and difluorocarbene to alkynes: a new active Pb-mediated approach to 2-fluoropyrrole derivatives. J. Chem. Soc., Perkin Trans. 1, 2000, 231-238, DOI: 10.1039/A905518E, http://pubs.rsc.org/en/Content/ArticleLanding/2000/P1/a905518e#!divAbstract
Международное сотрудничество:
Armin de Meijere (Göttingen University, Germany)
Andreas Hirsch (Friedrich-Alexander Universität Erlangen, Germany)
Kourosch Abbaspour Tehrani (Antwerp University, Belgium)
Heinz Heimgartner (Zurich University, Switzerland)
Grzegorz Mlostoń (University of Poznan, Poland)
Pedro J. Pérez (University of Huelva, Spain)
Фонды, гранты:
Saint Petersburg State University Grants
Russian Fundation for Basic Research
Ministry of Education Programs
Grants for Young Researchers from the President of Russia
DAAD (Germany)















